Engineered for Clinical Precision and Patient Safety
 Premium Organic Cotton Tampons - Eco-Friendly Disposable Hygiene Product
Premium Organic Cotton Tampons - Eco-Friendly Disposable Hygiene Product
 Factory Price Medical Fenestrated Surgical Drape with CE Certification
Factory Price Medical Fenestrated Surgical Drape with CE Certification
 3q Protective Customized Breathable Sofa Factory Covers Medical Boot Disposable Non Woven PP Shoe Cover
3q Protective Customized Breathable Sofa Factory Covers Medical Boot Disposable Non Woven PP Shoe Cover
 Professional Brand Protective Instrument Glass Disposable Microscope Cover
Professional Brand Protective Instrument Glass Disposable Microscope Cover
 Sterile Iodine Antimicrobial Incise Drapes Advanced Adhesive Surgical Film for Operating Room Infection Prevention
Sterile Iodine Antimicrobial Incise Drapes Advanced Adhesive Surgical Film for Operating Room Infection Prevention
 Highly Elastic Medical Spunbond Polypropylene Nonwoven for Hygienic Products
Highly Elastic Medical Spunbond Polypropylene Nonwoven for Hygienic Products
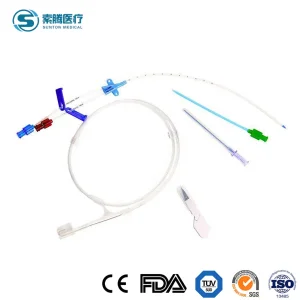 Sunton Manufacturer Price Surgical Sterile CVC Catheter Central Venous Catheterization Kit Wholesale Custom Cheap Price China Antimicrobial CVC Catheter Set
Sunton Manufacturer Price Surgical Sterile CVC Catheter Central Venous Catheterization Kit Wholesale Custom Cheap Price China Antimicrobial CVC Catheter Set
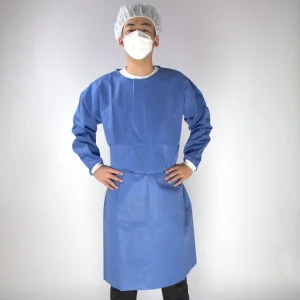 Sell Well Dust-Proof Surgical Gown 40GSM Blue Disposable SMS Non-Woven Isolation Gown
Sell Well Dust-Proof Surgical Gown 40GSM Blue Disposable SMS Non-Woven Isolation Gown
In the modern healthcare landscape, the Surgical Dressing Pack has evolved from a simple collection of consumables to a sophisticated tool of clinical efficiency. As a leading OEM/ODM Surgical Dressing Pack Factory, we understand that "Information Gain" in this sector is not just about raw materials; it's about the semantic integration of sterility, ergonomics, and logistical optimization.
Global surgery volumes are projected to increase by 25% over the next decade. This growth demands a surgical supply chain that is both resilient and highly specialized. Our role as a supplier goes beyond manufacturing; we are architects of infection control. By providing pre-assembled, procedure-specific kits, we reduce the cognitive load on surgical staff, minimize the risk of "never events," and ensure that every item—from the fenestrated drape to the antimicrobial film—meets the highest E-E-A-T standards (Experience, Expertise, Authoritativeness, and Trustworthiness).
The "Search Intent" of modern procurement officers has shifted from "cheap medical kits" to "validated surgical outcomes." This is where our expertise becomes a competitive advantage. We don't just supply products; we provide data-backed solutions that address the macro-economic pressures of hospital management, such as reducing turnover time in the Operating Room (OR) and lowering the incidence of Surgical Site Infections (SSIs).
Shanghai SwellMed Medical Co., Ltd. is a globally recognized manufacturer of high-quality single-use sterile medical devices, with a strong focus on infection control and surgical safety. Our core strength lies in seamlessly integrating cutting-edge R&D with practical clinical application.
We possess deep domain expertise and hold numerous patents for our innovative technologies. Our products, backed by robust clinical data, are designed with a user-centric approach, ensuring reliability, ease of use, and improved patient outcomes. Furthermore, our global compliance certifications (e.g., FDA, CE) and dedicated professional support team guarantee trust and partnership at every stage.

We leverage a decentralized logistics model to ensure that our OEM surgical packs reach healthcare providers in Europe, North America, and Asia without the bottlenecks of traditional medical supply chains.
Full adherence to MDR (EU) 2017/745 and FDA 510(k) requirements. Our quality management systems are ISO 13485 certified, providing a foundation of absolute reliability.
Our "Innovation-as-a-Service" model allows clinical partners to co-develop specialized dressing packs for robotic surgery, cardiology, and orthopedics, capturing specific market niches.
Transitioning from synthetic non-wovens to biodegradable organic cotton and plant-based polymers to reduce medical waste.
Integration of RFID and IoT sensors within procedure packs to track sterility shelf-life and inventory levels in real-time.
Utilizing AI-driven data mining to predict the specific consumable needs of individual surgeons, creating hyper-personalized ODM kits.
Developing "Close-the-Loop" recycling programs for used surgical drapes and gowns, converting waste back into raw industrial materials.
Optimized sterile kits designed for high-turnover outpatient procedures, where speed and consistency are paramount for operational profitability.
Vacuum-sealed, ruggedized dressing packs engineered for stability in extreme environments, providing military-grade infection prevention.
Specialized ODM packs featuring iodine-impregnated antimicrobial films and highly absorbent non-wovens for chronic wound management.
Supporting over 50,000 successful procedures worldwide with 200% growth in trust.










End-to-End Solutions for Modern Operating Rooms
 Medical Disposable Sterilized Surgical Drape Environmental Protection Pack
Medical Disposable Sterilized Surgical Drape Environmental Protection Pack
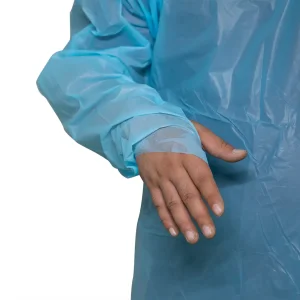 Standard Reinforced Disposable Surgical Non Woven Waterproof CPE Gown with Thumb Loop
Standard Reinforced Disposable Surgical Non Woven Waterproof CPE Gown with Thumb Loop
 Hospital White Disposable Waterproof SPA Massage Medical Examination Bed Sheet Cover for Hospital
Hospital White Disposable Waterproof SPA Massage Medical Examination Bed Sheet Cover for Hospital
 OEM Medical Consumables Wound Care/Circumcision Procedure Pack/Basic Sterile Surgical Drape Set Pack with CE13485
OEM Medical Consumables Wound Care/Circumcision Procedure Pack/Basic Sterile Surgical Drape Set Pack with CE13485
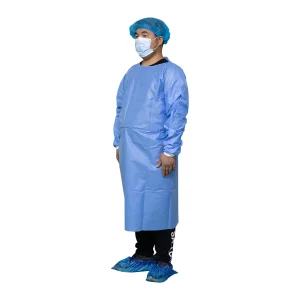 Disposable Isolation Gown Non Woven Gowns Waterproof PP+PE Isolation Gowns
Disposable Isolation Gown Non Woven Gowns Waterproof PP+PE Isolation Gowns
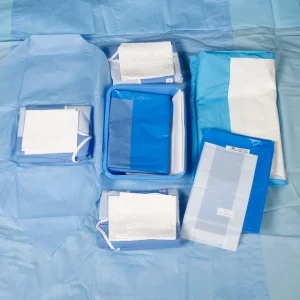 Hospital Operating Room OEM Sterile Surgical Pack Disposable Operation Kit Factory for Surgery Operation Usage
Hospital Operating Room OEM Sterile Surgical Pack Disposable Operation Kit Factory for Surgery Operation Usage
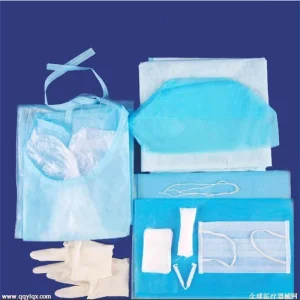 Cardiology Catheter Procedure Pack Cardiovascular Pack
Cardiology Catheter Procedure Pack Cardiovascular Pack
 Disposable Short Sleeve/Sleeveless Isolation Gown Non Woven SMS/PP Surgical Gown Medical Patient Gown Dark Blue for Dental Clinic/Hospital/Laboratory
Disposable Short Sleeve/Sleeveless Isolation Gown Non Woven SMS/PP Surgical Gown Medical Patient Gown Dark Blue for Dental Clinic/Hospital/Laboratory