⚙️The Evolution of Reusable Surgical Drapes: A Macro-Industry Analysis
In the contemporary healthcare landscape, the pivot toward sustainability without compromising clinical sterility is a paramount challenge. As a specialized OEM manufacturer and supplier, Shanghai SwellMed recognizes that the paradigm shift from single-use to high-performance reusable surgical drapes is driven by three core pillars: Environmental Stewardship (ESG), Cost-Efficiency in Hospital Operations, and Advanced Material Engineering.
Our research indicates that healthcare facilities worldwide are increasingly seeking "closed-loop" supply solutions. Reusable surgical drapes, engineered with advanced barrier fabrics (like high-density polyester laminates), offer superior breathability and fluid resistance compared to traditional disposables, ultimately reducing the carbon footprint of surgery centers while maintaining stringent AAMI Level 3 and 4 barrier requirements.
🔬Technical Roadmap & Material Innovation
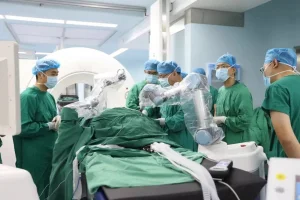
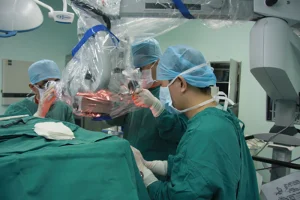
At the intersection of textile engineering and medical safety, our production line leverages cutting-edge fabrication techniques. We prioritize technical textiles that withstand repeated autoclaving (steam sterilization) and industrial washing cycles. Our R&D team continuously integrates hydrophobic coating technologies with ergonomic design principles. By analyzing the "Information Gain" of current surgical procedures—such as the specific anatomical draping requirements for robotic surgery or orthopedic procedures—we tailor our custom OEM solutions to minimize surgical setup time, thus optimizing throughput for operating rooms.
🌐Global Commercial Landscape & Localized Compliance
Navigating the global medical device market requires a profound understanding of localized regulatory frameworks. Shanghai SwellMed operates as a pivotal bridge, ensuring that our partners in the US, EU, and emerging markets receive products compliant with FDA 21 CFR Part 820, ISO 13485:2016, and the European Medical Device Regulation (MDR).
Compliance & Support Framework:
- Rigorous Sterility Validation: Adherence to ISO 11135 and ISO 17665 standards for sterilization assurance levels.
- Custom OEM/ODM Integration: We don't just supply products; we integrate into your clinical workflow, providing design documentation, clinical evaluation reports (CERs), and comprehensive technical files to expedite your local regulatory approvals.
- Regional Logistics: Optimized supply chain management designed to mitigate geopolitical risk and ensure just-in-time (JIT) delivery for high-volume hospital networks.
💬Expert Q&A (FAQ)
Q: How do reusable drapes compare to disposable in infection control?
A: Modern reusable drapes utilize advanced microbial barrier fabrics that are tested to match or exceed the performance of non-woven disposables. When processed in verified, controlled laundry facilities, they maintain consistent barrier efficacy over dozens of cycles.
Q: Can you handle custom specifications for OEM/ODM projects?
A: Absolutely. Our manufacturing facility specializes in custom geometry, specialized material laminates, and integrated device features (such as tubing organizers or fluid collection pouches) tailored to your specific surgical discipline.
Q: How does SwellMed ensure consistent quality across global orders?
A: We employ a strict Total Quality Management (TQM) system. Every batch undergoes physical, microbial, and chemical testing before shipment. We provide full traceability documentation for every serialized lot.
🏭Our Factory: Excellence in Manufacturing
Shanghai SwellMed Medical Co., Ltd. is a globally recognized manufacturer of high-quality sterile medical devices, with a strong focus on infection control and surgical safety. Our core strength lies in seamlessly integrating cutting-edge R&D with practical clinical application. We possess deep domain expertise and hold numerous patents for our innovative technologies. Our products, backed by robust clinical data, are designed with a user-centric approach, ensuring reliability, ease of use, and improved patient outcomes. Furthermore, our global compliance certifications (e.g., FDA, CE) and dedicated professional support team guarantee trust and partnership at every stage.

 SwellMed Medical
SwellMed Medical